Trevena Inc (NASDAQ:TRVN) is moving up a bit in Wednesday’s trading session. If it can clear yesterdays high a rally could be in the cards.
Market Action
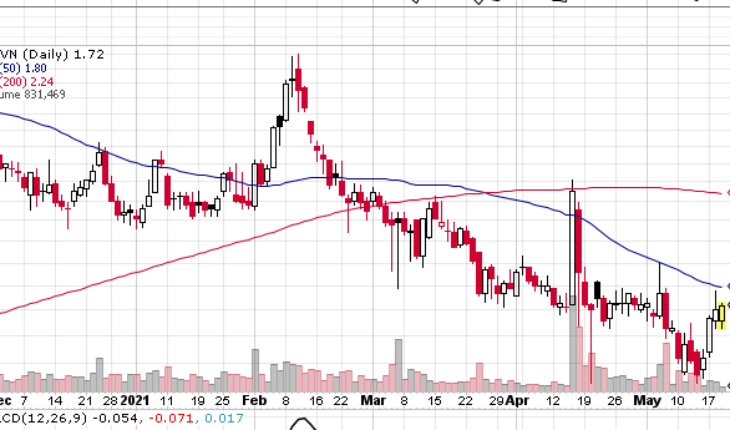
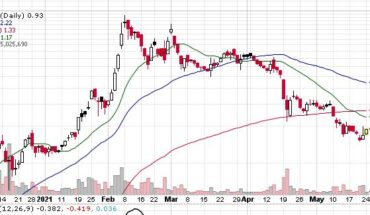
As of 12:39, TRVN stock has jumped 1.17% to $1.72. The stock has seen a total volume of 848K shares, compared to its average volume of 3.57 million shares. The stock has moved in range of $ 1.6300 – 1.7300 after opening at $1.66.
TRVN Releases News Today
Trevena Announces Presentations of OLINVYK® Health Economic Models at ISPOR 2021
- TRVN today announced two poster presentations at the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) 2021 Annual Conference. The conference is being held virtually from May 17th to 20th, 2021.
- The posters highlight two health economic models developed for OLINVYK (oliceridine) injection, which estimate the budget impact of OLINVYK compared to IV morphine when used on-demand in a hospital setting for postoperative pain.
- Both models were also presented at the Academy of Managed Care Pharmacy (AMCP) 2021 Annual Meeting.
- The base case model calculates a ~$230,000 decrease in total cost of care per 1,000 patients associated with OLINVYK, compared to IV morphine.
- When limited to patients who are both elderly (≥ 65 years old) and obese (BMI ≥ 30 kg/m2), the expected savings associated with OLINVYK increase to ~$364,000 per 1,000 patients, with the total cost of care associated with IV morphine exceeding $1.25M per 1,000 patients.
- These cost savings are due to reduced AEs for OLINVYK-treated patients, as observed in the Phase 3 pivotal trials, and using a conservative, low-end estimate of AE costs based on government and published literature sources.
“I am pleased to have another opportunity to present our compelling OLINVYK health economic data at a major medical conference,” said Mark A. Demitrack, M.D., Senior Vice President and Chief Medical Officer of Trevena, Inc. “Both models have been well received by hospital decision makers, who are using the cost offset and budget impact information from these models to support their formulary review of OLINVYK.”