Soleno Therapeutics (NASDAQ:SLNO) is the notable gainer and trading with major rally in the pre-market session after the news this morning.
Soleno Therapeutics Receives Orphan Drug Designation from FDA for Diazoxide Choline for the Treatment of Glycogen Storage Disease Type 1a
- announced that the U.S. Food and Drug Administration (FDA) has granted its investigational drug, DCCR (diazoxide choline) Extended-Release tablets, Orphan Drug Designation for the treatment of Glycogen Storage Disease Type 1a (GSD 1a), or von Gierke disease. DCCR previously received Orphan Drug Designation for the treatment of Prader-Willi syndrome.
- The FDA’s Office of Orphan Drug Products grants Orphan Drug Designation to support drug candidates in development for underserved patient populations or rare disorders that affect fewer than 200,000 people in the United States. Orphan Drug Designation qualifies a candidate for various development incentives, including tax credits for eligible clinical trials, waiver of application fees and market exclusivity for seven years upon FDA approval.
“The granting of Orphan Drug Designation for our DCCR program in an additional indication, GSD 1a, represents a significant milestone for Soleno,” said Anish Bhatnagar, M.D., Chief Executive Officer of Soleno Therapeutics. “GSD 1a is a type of glycogen storage disease marked by the body’s inability to metabolize glycogen into glucose, resulting in hypoglycemia, high levels of fat in the blood, and impaired growth, among other complications. We believe DCCR’s mechanism of action as an ATP-dependent potassium channel agonist, with the potential to regulate hypoglycemia and reduce fatty acid synthesis, could provide a meaningful treatment option for GSD 1a, a condition for which there are currently no approved therapies.”
Market Action
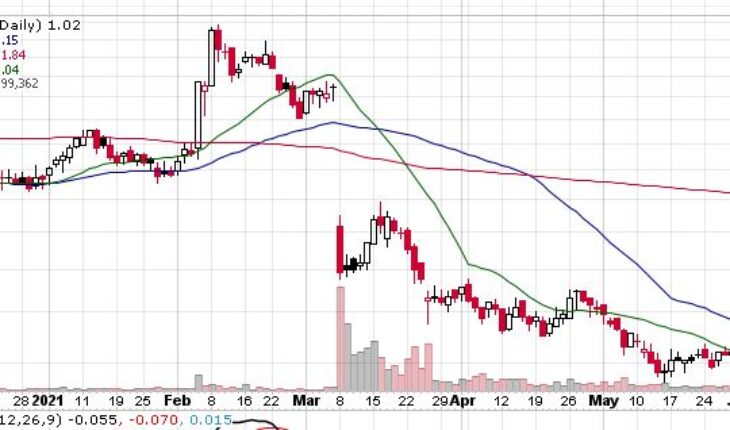
As of 8:30, SLNO stock soared 19 cents or 18.63% to $1.21. So far the stock has traded 1.66 milion shares traded hands.