Vascular Biogenics Ltd (NASDAQ:VBLT) is trading with moderate gains in the pre-market session after the news on Thursday.
VBL Therapeutics Announces Addition of Progression Free Survival as a Second Primary Endpoint in OVAL, a Phase 3 Registration Enabling Study of VB-111 in Ovarian Cancer
- announced a primary endpoint amendment in the OVAL Phase 3 registration-enabling study of VB-111. The clinical trial amendment included a second, separate primary endpoint, of progression free survival (PFS), in addition to the original primary endpoint of the trial, overall survival (OS).
- Based upon the changes that were reviewed by the U.S. Food and Drug Administration (FDA), successfully meeting either primary endpoint is expected to be sufficient to support BLA submission. Successful meeting of the PFS endpoint, with a readout anticipated in 2022, could accelerate BLA submission by approximately one year compared to original projections based on the readout of the OS primary endpoint that remains anticipated in 2023.
- The OVAL study amendment, along with an update on the number of patients enrolled, which as of April 30, 2021, exceeded 260 patients, will be presented tomorrow as part of a virtual Clinical Trial in Progress poster presentation at the 2021 ASCO Annual Meeting.
Market Action
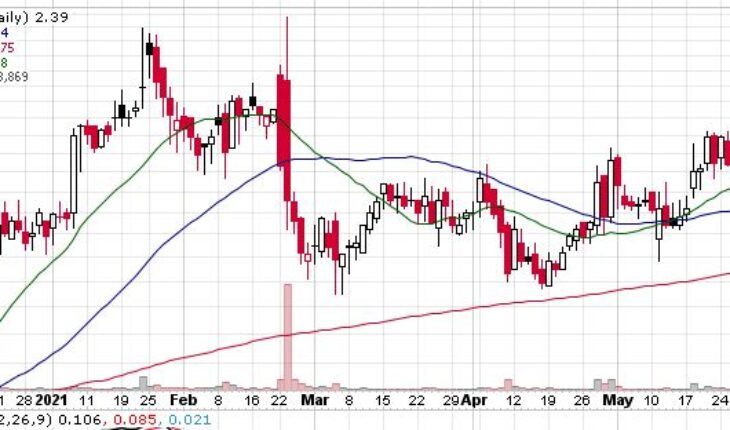
As of 8:55, VBLT stock jumped $0.21 or 8.79% to $2.60. So far the stock has traded 377K shares traded hands.