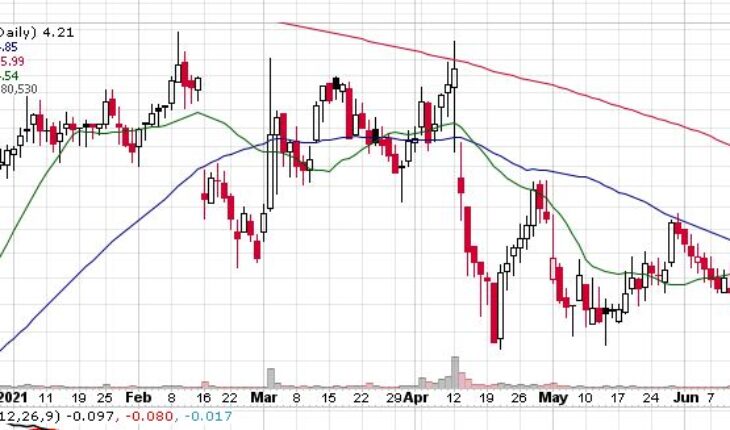
Avenue Therapeutics Inc (NASDAQ:ATXI) is the biggest loser in the pre-market as investors are reacting negatively to the company data from FDA.
Major Trigger:
Avenue Therapeutics Receives Complete Response Letter from the FDA for IV Tramadol
Key Highlights:
- announced that it has received a second Complete Response Letter (“CRL”) from the U.S. Food and Drug Administration (“FDA”) regarding its New Drug Application (“NDA”) seeking approval for IV tramadol.
- The CRL stated that the delayed and unpredictable onset of analgesia with IV tramadol does not support its benefit as a monotherapy to treat patients in acute pain, and there is insufficient information to support that IV tramadol in combination with other analgesics is safe and effective for the intended patient population. The FDA did not identify any Chemistry, Manufacturing and Controls (“CMC”) issues in this CRL.
- Avenue disagrees with the FDA’s interpretation of the data in the NDA and intends to continue to pursue regulatory approval for IV tramadol.
Market Reaction
As of 7.45, ATXI stock slumped 36% to $2.70 with 105K shares traded hands.