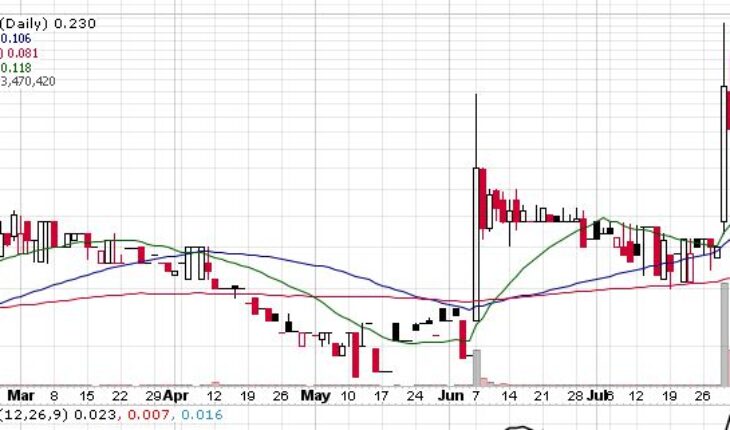
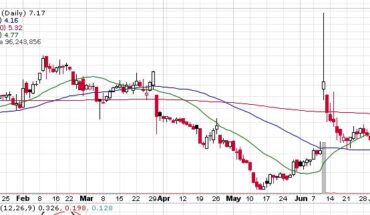
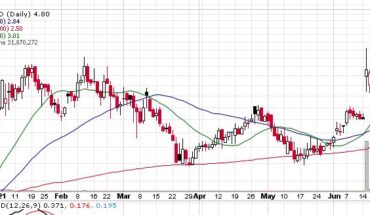
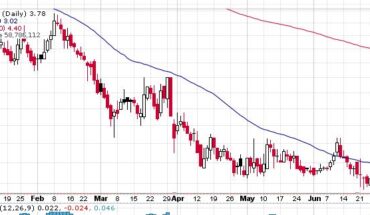
Clinical stage biopharmaceutical company, Adhera Therapeutics (OTCMKTS:ATRX) on 29th July, 2021 announced a letter of intent dated June 7, 2021 executing definitive license agreement with Melior Pharmaceuticals for which Adhera will be licensing MLR-1019 from Melior Pharma 2, for initial purpose of developing fresh therapeutic for Parkinson’s disease.
The company believes leveraging safety data done by Melior Discovery will qualify advancing MLR-1019 directly into a Phase 2a clinical trial for PD.
Andrew Kucharchuk, Chief Executive Officer at Adhera, said that MLR-1019 shows that it can be a big step in treating Parkinson’s disease for providing patients relief for both non-movement and movement symptoms. Kucharchuk said that it is excited on having Melior group of companies as partners and collaborators on the project and assessing MLR-1019 for delivering safe option to patients and neurologists for managing the disease.
Andrew Reaume, CEO of Melior, said it is having a shared vision towards armesocarb as innovative new therapy for addressing Parkinson’s disease. Reaume said that complete set of global resources will deepen the relationship.
Armesocarb, an active enantiomer of the racemic mixture mesocarb, is highly selective dopamine approved in Soviet Union in 1971. Melior discovered benefits of MLR-1019 for PD, securing the intellectual property with several patents.