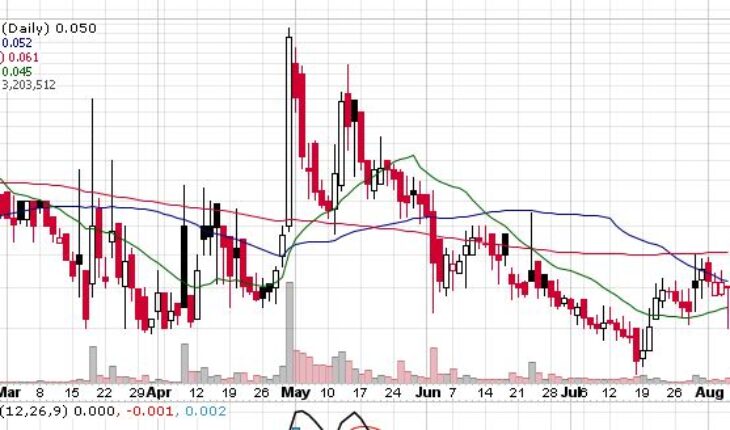
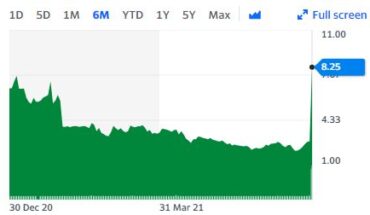
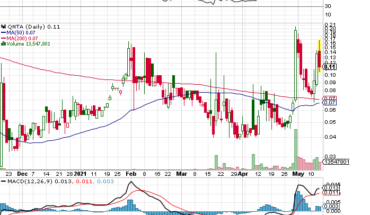
Global integrated biomedical firm with R&D, manufacturing as well as consumer product distribution, Medolife Rx, a subsidiary of Quanta Inc (OTCMKTS:QNTA), on August 5, 2021, announced the launching of a pharmacokinetic study on its lead drug candidate Escozine.
The study will aim to give a basis for assessing drug exposures in the body. Requested by the United States Food and Drug Administration for its Pre-Investigational New Drug filing, Escozineis potential therapeutic for the Covid-19 virus.
Medolife is doing PK as well as a biodistribution study of Escozine in rodents, being done in a lab in the UK, which is also a requirement of the FDA.
Medolife CEO Dr. Arthur Mikaelian said that additional PK study is the next step in the company’s FDA IND submission process and vital for giving the data needed for formulating therapeutic dosing needed for eventual human trials.
Mikaelian added that the drug has demonstrated effectiveness in pre-clinical proof-of-concept trials done so far to take it further into Phase II clinical trials.
The company also announced joining the highly anticipated “Tony Hawk’s Vert Alert” competition, which will feature the most elite current skaters and returning Olympians.
To be conducted at Utah State Fairgrounds in Salt Lake City on August 27 and 28, this is the first major audience-attended skateboarding competition since the start of the pandemic, which will attract more than 8000 attendees over the two days of competition.
They will also be the firm’s public introduction of the Immunapen brand, which utilizes the firm’s proprietary polarized scorpion peptide formulation.
The company had signed a product endorsement in June with Tony Hawk, to promote groundbreaking cannabis-based products.