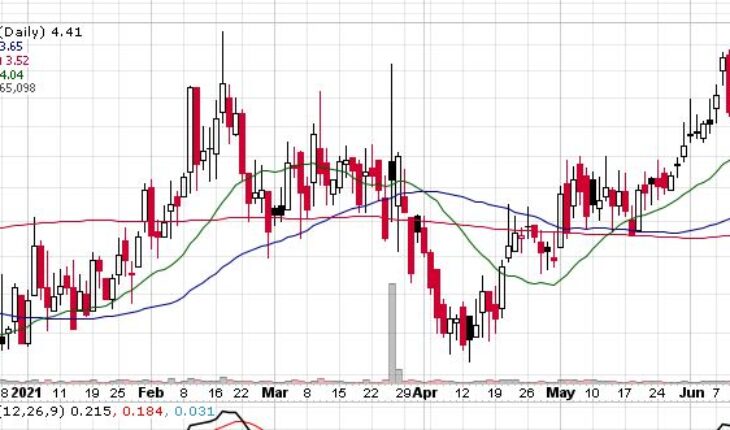
Enochian Biosciences Inc (NASDAQ:ENOB) is moving higher again in the pre-market session on Wednesday, extending its monthly gain to 20%.
Major Trigger:
Enochian BioSciences Announces FDA Acceptance of Pre-IND Request For Potential HIV Cure
Key Highlights:
- today announced that the FDA has accepted a Pre-IND (Investigational New Drug) request for a potential functional cure or treatment of HIV. Written comments are expected this Fall.
- Dr. Serhat Gumrukçu, co-founder and inventor of Enochian BioSciences, and Director of Seraph Research Institute (SRI), submitted the Pre-IND. The request was based on the results of a 54-year old man living with HIV who had failed to suppress the virus with antiviral therapy.
- The patient subsequently achieved viral control for 255 days with an innovative treatment of Natural Killer (NK) and Gamma Delta T-cells (GDT) collected from another person. During the entire period, no antiviral drugs were given. It is believed that the GDT cells, a small subset of immune cells that can be infected with HIV, could be a key factor in controlling the virus.
- This innovative cellular therapy could be an important approach to achieve a “functional cure” of HIV, potentially allowing persons with the virus to stop antiviral treatment for extended periods of time. The Pre-IND submission requested that the novel strategy be extended to persons with HIV who have achieved suppression of the virus with antiviral treatment.
Market Reaction
As of 7:40, ENOB stock jumped 6.35% to $4.69. So far the stock has traded 663K shares traded hands.